Shock wave application to cell cultures
However, this question remains: What happens to the waves after passing the cell culture? The difference of the acoustic impedance of the cell culture medium and the ambient air is that high, that more than 99% of shock waves get reflected! We therefore developed a model that mainly consists of a Plexiglas built container that allows the waves to propagate in water after passing the cell culture. This avoids cavitation effects as well as reflection of the waves that would otherwise disturb upcoming ones. With this model we are able to mimic in vivo conditions and thereby gain more and more knowledge about how the physical stimulus of shock waves gets translated into a biological cell signal (“mechanotransduction”).
Introduction
Shock waves are sound pressure waves arising from a sudden release of energy, e.g. as thunder when lightning. In medicine shock waves have been used for over 30 years in Lithotripsy for the disintegration of kidney stones. Since the incidental finding of iliac bone thickening in Lithotripsy patients in the early 1980s, first studies were conducted to evaluate the effect of shock wave treatment (SWT) on bone healing1. Impressive results of improved healing of long-bone nonunions could be observed2. Subsequently, indications were expanded to soft tissue wounds3. Basic research findings showed that shock waves do cause a biological stimulus to the target tissue without any subsequent damage. Release of angiogenic growth factors (e.g. VEGF, PlGF, FGF) is followed by significant angiogenesis. This led to a further expansion of indications towards ischemic pathologies. Our group and others showed the positive effect of SWT on ischemic heart disease in animal models as well as in clinical trials4-6.
However, the exact mechanism of how the physical stimulus of SWT is translated into a biological signal (mechanotransduction) remains largely unknown. As the interest in SWT from several fields of medicine increases continuously, the quest for the mechanism is getting more and more intense. Therefore, in vitro shock wave experiments are gaining importance. Besides reduction of animal experiments and cost-effectiveness, the biggest advantage of in vitro shock wave treatment (IVSWT) may be the possibility of studying the specific behavior of a certain cell type. In shock wave mediated tissue regeneration most likely all cells of the treated tissue are involved, even systemic effects are discussed. Nevertheless, each cell type plays a specific role and has its own intrinsic function. IVSWT enables us to detect this particular function and thereby gives us better understanding of the complex underlying processes.
Today’s knowledge about shock wave’s effects on cell cultures includes the increase of proliferation, alteration of cell membrane receptors, increase and acceleration of cell differentiation, release of growth factors and chemo-attractants as well as increased cell migration7-9.
Distracting physical effects in most in vitro models Various methods of applying shock waves onto cell cultures have been described. This fact leads to the problem that it is highly difficult to compare results, as physical conditions of cell stimulation are quite different between these models. In general, all existing models focus on how to best apply shock waves onto cells.
However, this question remains: What happens to the waves after passing the cell culture? The main problem is that the difference of the acoustic impedance of the cell culture medium and the ambient air is that high, that more than 99% of shock waves get reflected Figure 1.
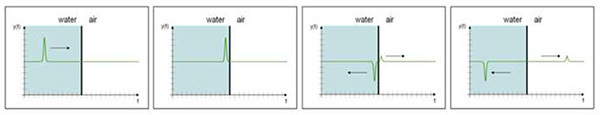
Due to the difference in acoustic impedance of the two media the waves are not only reflected but a phase-shift of 180° occurs resulting in strong tensile forces to the cells Figure 2.
Acoustic impedance is defined as the product of the density of a material and its sound velocity Z=ρ x c. For water the acoustic impedance is ZWater= 1,440,000 Ns/m3, for air it is only 420 Ns/m3. The large difference of these two values results in reflection and phase shift of shock waves. The phase shift turns a positive pressure pulse into a tensile wave.
Even if this tensile force is not harmful to the cells, it interferes with the idea of mimicking in vivo shock wave effects in vitro. In vivo these tensile forces hardly occur due to large body structures.
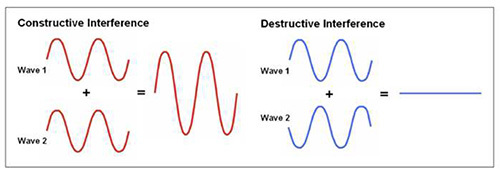
Furthermore, the back running waves can even disturb the incoming ones. This may cause interference. Two types of interference are known. Constructive interference means that both waves are added thereby resulting in doubled amplitude Figure 3. Destructive interference occurs if waves meet diametrically opposed. It causes abolishment of waves (Figure 3). Therefore, IVSWT needs a model that enables shock waves to propagate after passing the cell culture.
IVSWT water bath
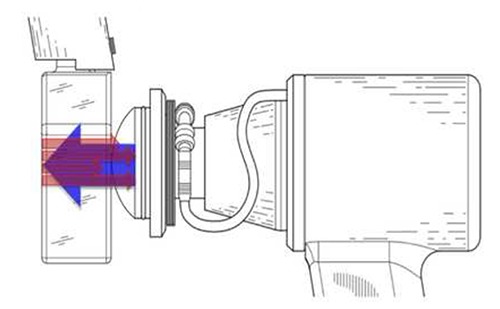
Considerations following the above mentioned concerns lead us to design a water bath for avoiding the described problems Figure 4. Basically, it consists of a Plexiglas built container with a membrane to connect every kind of shock wave applicator. For coupling between this membrane and the applicator ultrasound transmission gel has to be used. The water bath is filled with degassed water to avoid cavitation that would occur if gas was soluted within the water. A heater at the bottom with a temperature sensor connected to a control unit enables to regulate temperature for imitation of in vivo conditions and to avoid cell cultures to cool down during the procedure. Temperature can be held stable at 37 degrees centigrade as it is done in an incubator. A holder for the cell samples allows for immersing any kind of culture flask or tube. Thereby, the sample vessel needs to be completely filled with culture medium, as air bubbles would block shock waves! A wedge shaped absorber at the back wall of the bath destructs waves in order not to get reflected and run back to avoid interference.
A further advantage to other IVSWT models is the possibility of varying the distance between the applicator and the culture flasks. Findings of our group and others who use this model clearly show that every cell type reacts very specifically to different treatment parameters. Moreover, defining the distance between the source of the waves and the sample is crucial as it enables us to control the cells to be at a specific position in relation to the focus of the shock wave applicator.
Protocol
Ethical permission
After obtaining written informed consent of patients, umbilical cords were obtained from Caesarean section at the Department of Gynaecology for isolation of human umbilical vein endothelial cells (HUVECs). Permission was given from the ethics committee of Innsbruck Medical University (no. UN4435).
1. Prepare the IVSWT Water Bath
-
Prepare 3.5 L of tap water in an appropriate tank. Water needs to be heated to 37 °C (see Protocol 6).
-
Fill the water into the water bath until the water level is approximately 3 cm below the edge. Mind to completely cover the membrane with water. This will require approximately the prepared 3.5 L.
-
Connect the temperature sensor to the power supply.
-
Put the sensor into the intended fitting on the back wall of the water bath.
-
Connect heating to power supply of the temperature sensor. Do not do this without water in the bath as the heater would melt the Plexiglas.
-
Wait until the water has reached a stable temperature of 37 °C. Stir the water regularly with some kind of stick to guarantee constant temperature throughout the water bath.
2. Prepare Cells and Culture Flasks
-
Seed cells over night at desired density in T25 culture flasks or culture the cells in the flasks directly until they have reached the desired confluence.
-
Before the experiment, align the flasks vertically.
-
Fill the culture flasks with culture medium until right below their necks. Do not fill too much medium into the flasks as medium in contact to the neck or the closing cap would get contaminated.
-
Screw the flasks with solid caps without filters to prevent contamination from water. Mind that the water in the bath and the bath itself are not sterile.
-
Seal the caps with Parafilm before insertion into the water bath.
-
Fix sealed culture flasks in the provided stand.
-
Insert fixed flask into the water bath. Take care that the middle of the cell culture flask is at the same height as the center of the shock wave applicator’s membrane. A line at the side of the bath guides you.
3. Define Treatment Parameters
-
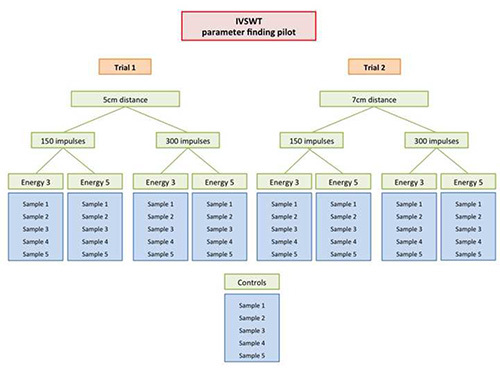
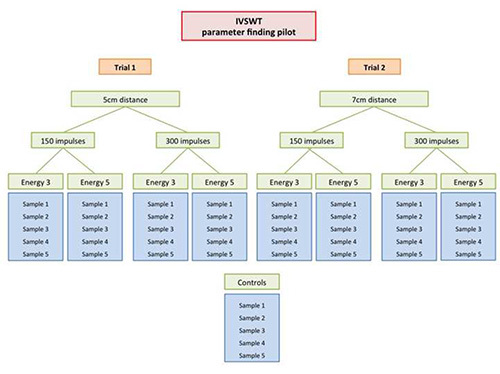
Define distance between shock wave source and sample. Identify perfect treatment parameters by conducting the parameter finding pilot experiment as described in Figure 6.
-
Choose the right treatment parameters (energy flux density, frequency) on the shock wave device. Again, refer to Figure 6.
4. Shock Wave Application
-
Put plenty amounts of commercially available ultrasound transmission gel on the shock wave applicator as well as on the membrane of the water bath. This is to assure coupling. No air or air bubbles should be between the applicator and the membrane as it would absorb shock waves.
-
Connect applicator to the membrane and hold it stable in the center of the membrane. Take care that it is aligned horizontally.
-
Make sure that the correct vertical position of the probe inside the water bath is in line with the indicated marking at the side of the bath.
-
Hold centers of the applicator and the flask stable in a horizontal line.
-
Activate the shock wave device and apply the impulses while keeping the culture flask as well as the applicator in a stable position during the whole procedure.
5. After Treatment
-
Take the culture flask out of the water bath.
-
Dry it with common paper towels.
-
Wipe the flask accurately with disinfecting agents.
-
Remove Parafilm sealing and cap.
-
Pipette the Medium inside the flasks into centrifugal tubes.
-
Centrifuge appropriately. Centrifuging parameters depend on the used cell type, e.g. for HUVECs, use 290 g at room temperature.
-
Add 5ml of cell culture medium to the flask. Resuspend the centrifuged cell pellet with 2 ml of cell culture medium. Pipette the suspension into the flask. Thus, cell fragments or cells that may have been detached during treatment do not get lost.
Pitfalls
-
Preparation as well as treatment should be carried out under sterile conditions inside laminar flow to avoid contamination of the cell culture.
-
Good cell culture practice is recommended to avoid contamination of the probes. In particular disinfection of the outside of the flasks is strongly recommended before putting them back to the incubator. The water bath, as well as the water inside, is not sterile!
-
Do not connect the heater to the power supply unless the water bath is filled with water to avoid damage to the Plexiglas built bath.
-
Use generous amounts of ultrasound gel on the shock wave applicator to guarantee proper coupling and wave propagation into the bath. Air and even small air bubbles do absorb shock waves!
-
Check position of the probes inside the water bath in relation to the applicator to ensure treatment of the entire growth area.
-
Do not dunk probes too deep into the water to avoid contamination.
Representative Results
Using the described method we applied shock waves to human umbilical vein endothelial cells (HUVECs) that we afore isolated from umbilical cords. Umbilical cords were obtained from elective caesarian sections.
HUVECs were treated at a confluence of 90% in a T25 cell culture flask with an electrohydraulic shock wave therapy system. Treatment parameters were an energy flux density of 0.1 mJ/mm2 and a frequency of 5 Hz. 300 impulses were applied from a distance of 5 cm from the shock wave source to the cell layer.
Gene expression of Tie-2 (Tyrosine kinase with immunoglobulin-like and EGF-like domains 2) mRNA was measured by real-time PCR analysis. It was significantly up-regulated over time (2 hr post SWT: 156.75±14.49, 4 hr: 141.03±9.71, 6 hr: 166.68±2.15; p<0.05 vs. CTR: 100.03±7.5) Figure 5B. This angiopoietin receptor gives a direct hint for endothelial cell proliferation and is an indicator for their ability towards angiogenesis. As a positive control we used Poly (I:C) (polyinosinic polycytidylic acid) that as an RNA structure analogue is known to stimulate endothelial cells (2 hr post SWT: 125.7±10.08, 4 hr: 191.73±5.15, 6 hr: 400.93±19.62; p<0.05 vs. CTR: 103.65±6.18) Figure 5A.
A cytokine array was performed 48 hours after treatment to analyze main inflammatory cytokines IL-6 (59.97±1.24, p<0.05 vs. CTR 19.00±0.44) and IL-8 (71.89±1.52, p<0.05 vs. CTR 29.50±0.87) that are substantially involved in early angiogenesis. Both were significantly increased in the treatment group as shown by the cytokine array Figure 5C and after quantification Figure 5D.

Discussion
The significance of the proposed model for in vitro shock wave treatment is the fact that waves can propagate after passing the cell culture in contrast to existing models. Thereby, disturbing physical effects such as tensile forces can be avoided. The model more closely resembles in vivo conditions than that by others applying waves to their cell culture flasks directly.
An additional advantage is the possibility of varying the distance between shock wave source and cells. This would not be possible if treating a culture flask directly. However, in vivo there certain differences in the distance between the applicator and the target area, depending on how deep inside the target tissue is. At the same time one can study the effect of cells being in specific positions to the wave’s focus point.
Conducting first experiments
According to our experience with cardiomyocytes, endothelial cells, fibroblasts and cells, every cell type needs its specific treatment parameters. We therefore strongly recommend a pilot trial for assessment of the adequate treatment parameters before conducting the desired experiments. This pilot trial needs to include different distances between the shock wave applicator and the sample as well as different energy flux densities. Also the most appropriate number of pulses should be established. The frequency of applying shock waves is still a matter of concern – even in vivo. A suitable protocol for a treatment parameter finding pilot trial is provided as a supplement Figure 6.
The use of the ideal culture vessel is of high importance for the success of the experiment. We prefer common cell culture flasks of any size. However, there may be reasons to use other vessels – e.g. to need smaller volumes for completely filling the vessel during shock wave application. Suitable materials from an acoustic point of view are PE (Z=1’760’000 Ns/m3), soft-rubber (Z=1’270’000 Ns/m3), polyamid (Z=1’960’000 Ns/m3). Less ideal is PVC (3’270’000 Ns/m3) Plexiglas (Z=3’260’000 Ns/m3) delrin (3’450’000 Ns/m3) polycarbonate (2’770’000 Ns/m3) or polypropylene (2’400’000 Ns/m3). Hard materials such as glass or metal shall not be used.
Critical steps
Defining the distance between the shock wave source and the sample can be quite tricky. The reason is that the source (e.g. electrode tips in an electro-hydraulic system) is located inside the applicator. Therefore, it is necessary to know the distance from the source to the applicators mantle. You may have to ask the manufacturer of the shock wave device.
A disadvantage of this model is the fact that the cell culture vial needs to be filled with culture medium. Filling e.g. cell culture flasks implies an increased and expensive consumption of culture medium. Besides costs, this fact also means that whatever the cells secrete is strongly diluted. Detecting molecules therefore gets much more difficult because of a decreased concentration. Hence, we started to use protein filters for centrifugation and thereby increase concentration again.
Besides the use of different types of cell culture vials as described above, the main modification for IVSWT is its use for tissue culture and ex vivo models. In particular, our group tested it with perfect results in the aortic ring angiogenesis assay10. This means that it is possible to hold any kind of organ tissue or tissue engineered grafts into the IVSWT water bath. Ex vivo shock wave application therefore in future could be used to improve different kind of cell seeding methods on engineered grafts.
Disclosures
The authors have nothing to disclose.
Acknowledgments
The authors thank Reiner Schultheiss and Wolfgang Schaden for their inspiration for this model. We also thank Christian Dorfmüller for his all time tremendous efforts to support our research.
Many thanks to Robert Göschl and Hans Hohenegger for careful technical realization of our ideas!
References
- Haupt G, Haupt A, Ekkernkamp A, Gerety B, Chvapil M. Influence of shock waves on fracture healing. Urology. 1992;39:529–532. [PubMed] [Google Scholar]
- Schaden W, Fischer A, Sailler A. Extracorporeal shock wave therapy of nonunion or delayed osseous union. Clin. Orthop. Relat. Res. 2001;387:90–94. [PubMed] [Google Scholar]
- Schaden W, et al. Shock wave therapy for acute and chronic soft tissue wounds: a feasibility study. J. Surg. Res. 2007;143:1–12. [PubMed] [Google Scholar]
- Tepeköylü C, et al. Shock wave treatment induces angiogenesis and mobilizes endogenous CD31/CD34-positive endothelial cells in a hindlimb ischemia model: Implications for angiogenesis and vasculogenesis. J. Thorac. Cardiovasc. Surg. 2013;146:971–978. [PubMed] [Google Scholar]
- Nishida T, et al. Extracorporeal cardiac shock wave therapy markedly ameliorates ischemia-induced myocardial dysfunction in pigs in vivo. Circulation. 2004;110:3055–3061. [PubMed] [Google Scholar]
- Fukumoto Y, et al. Extracorporeal cardiac shock wave therapy ameliorates myocardial ischemia in patients with severe coronary artery disease. Coron. Artery Dis. 2006;17:63–70. [PubMed] [Google Scholar]
- Gotte G, Amelio E, Russo S, Marlinghaus E, Musci G, Suzuki H. Short-time non-enzymatic nitric oxide synthesis from L-arginine and hydrogen peroxide induced by shock waves treatment. FEBS Lett. 2002;520:153–155. [PubMed] [Google Scholar]
- Wang FS, Wang CJ, Huang HJ, Chung H, Chen RF, Yang KD. Physical shock wave mediates membrane hyperpolarization and Ras activation for osteogenesis in human bone marrow stromal cells. Biochem. Biophys. Res. Commun. 2001;287:648–655. [PubMed] [Google Scholar]
- Mittermayr R, et al. Extracorporeal shock wave therapy (ESWT) minimizes ischemic tissue necrosis irrespective of application time and promotes tissue revascularization by stimulating angiogenesis. Ann. Surg. 2011;253:1024–1032. [PubMed] [Google Scholar]
- Baker M, et al. Use of the mouse aortic ring assay to study angiogenesis. Nat. Protoc. 2011;22:89–104. [PubMed] [Google Scholar]
Johannes Holfeld, 1 Can Tepeköylü, 1 Radoslaw Kozaryn, 1 Wolfgang Mathes, 1 Michael Grimm, 1 and Patrick Paulus 2
Author informationCopyright and License informationDisclaimer
